New microscope filters
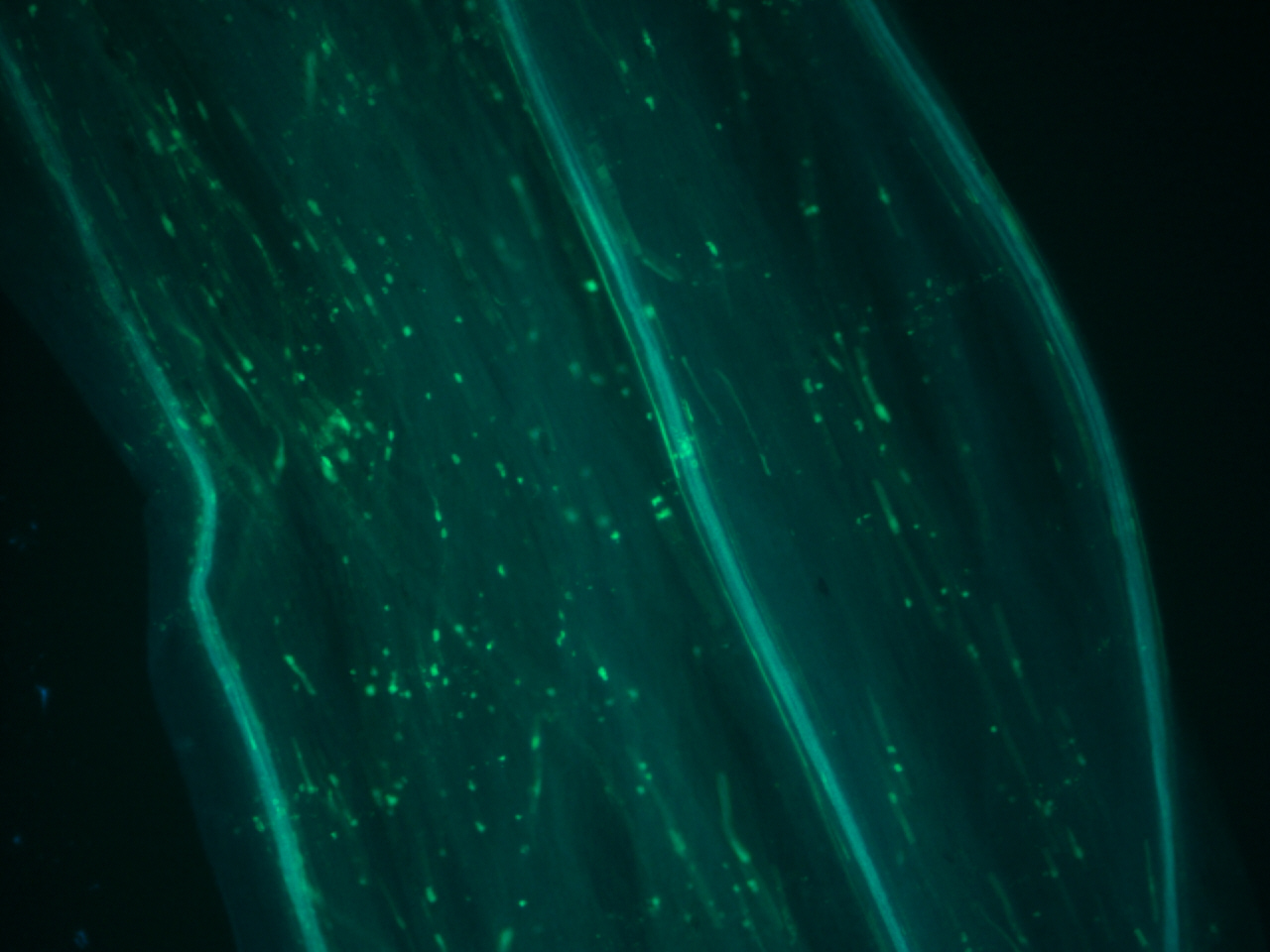
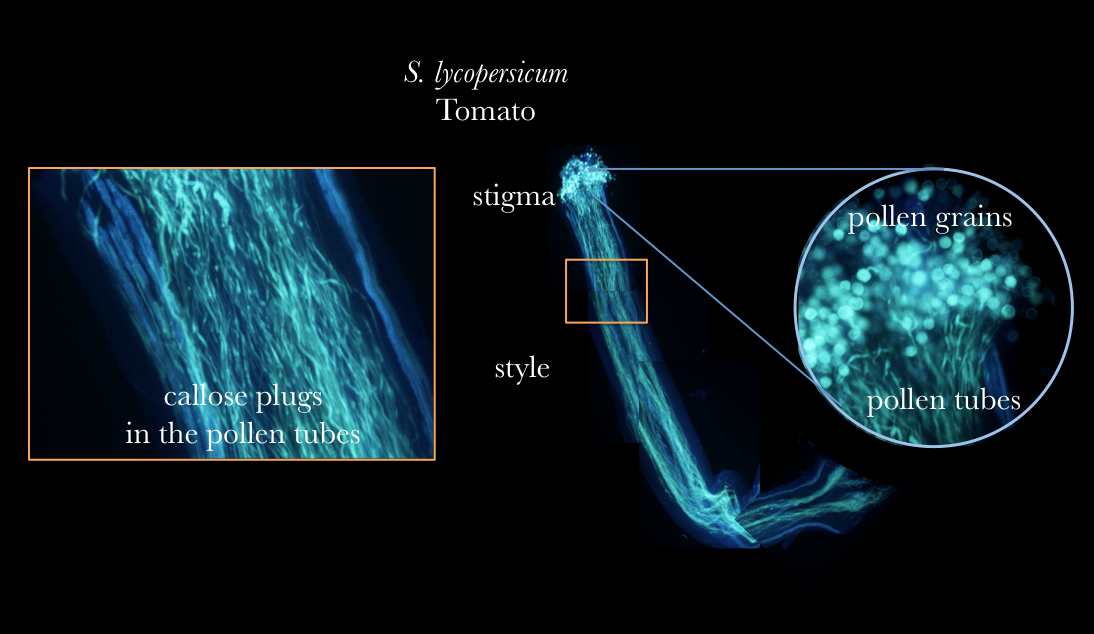
Pollen tubes stained with aniline blue
Generally I am more interested in vegetative development, but recently I have been paying a bit more attention to the part of plants that do not need any more flattery or attention - flowers. One of my recent projects made me interested in how tomato pollination occurs. This lead me to look at what happens when pollen hits sigma, the foreplay on the road to fertilization, yes sex. I have been very excited this week as we just recieved a few new filters on our dissecting microscope allowing visualization of pollen germination directly on the plant in the most beautiful way.
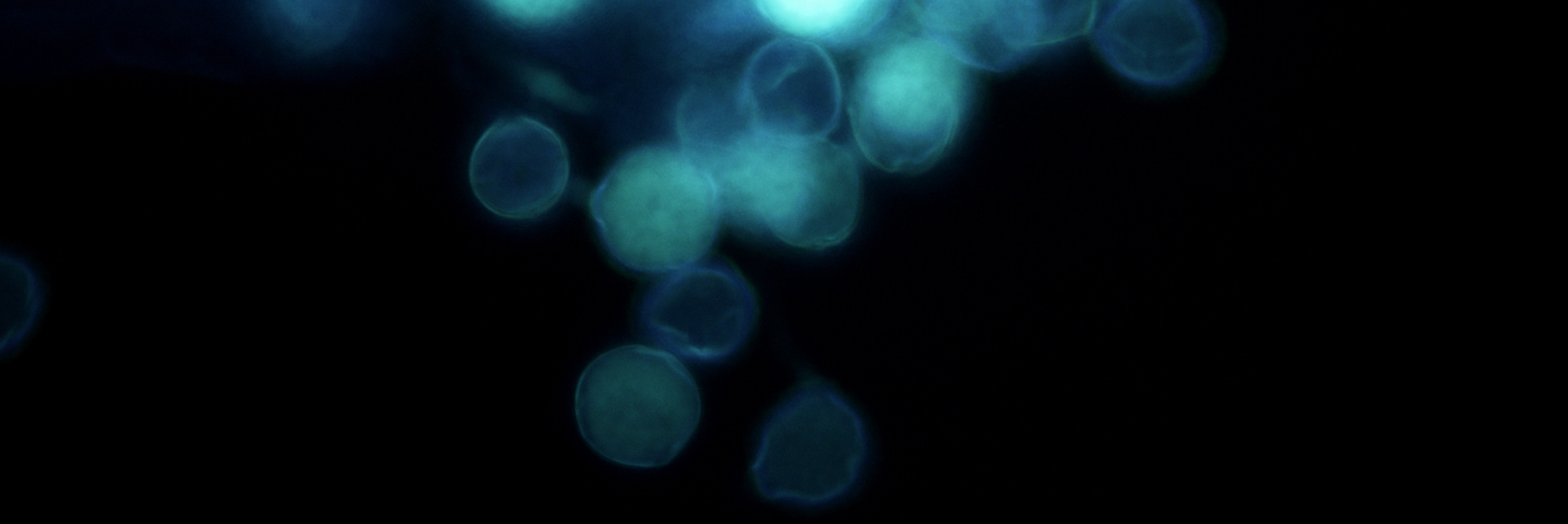
Pollen
Pollen grains come from the male part of a flower, the anther. When the pollen grain comes into contact with the female stigma, the pollen grain begins to elongate, creating the pollen tube. The pollen tube travels down the style where it will finally reach the ovary and this is when fertilization occurs. One of the most fascinating aspects of pollen tube development is that the pollen tube is just a single cell. The pollen tube does not grow by cellular division, but rather elongation. Many plant scientists study pollen tubes as a system because the pollen tube 1. length is so long compared to other plant cells 2. grows at such a rapid pace and 3. the polar (one side of the cell) nature of the growth.
The aniline blue is staining the callose in the pollen tubes. Pollen tubes often have callose plugs, which are the spots you see in the diagram. In all flower plants, pollen tubes form pollen plugs. They are believed to act by cutting the older parts of the pollen tube from the newly growing tip, possible increasing growth rate of the pollen tube down to the ovaries, but it is still largely unknown the exact biological function.
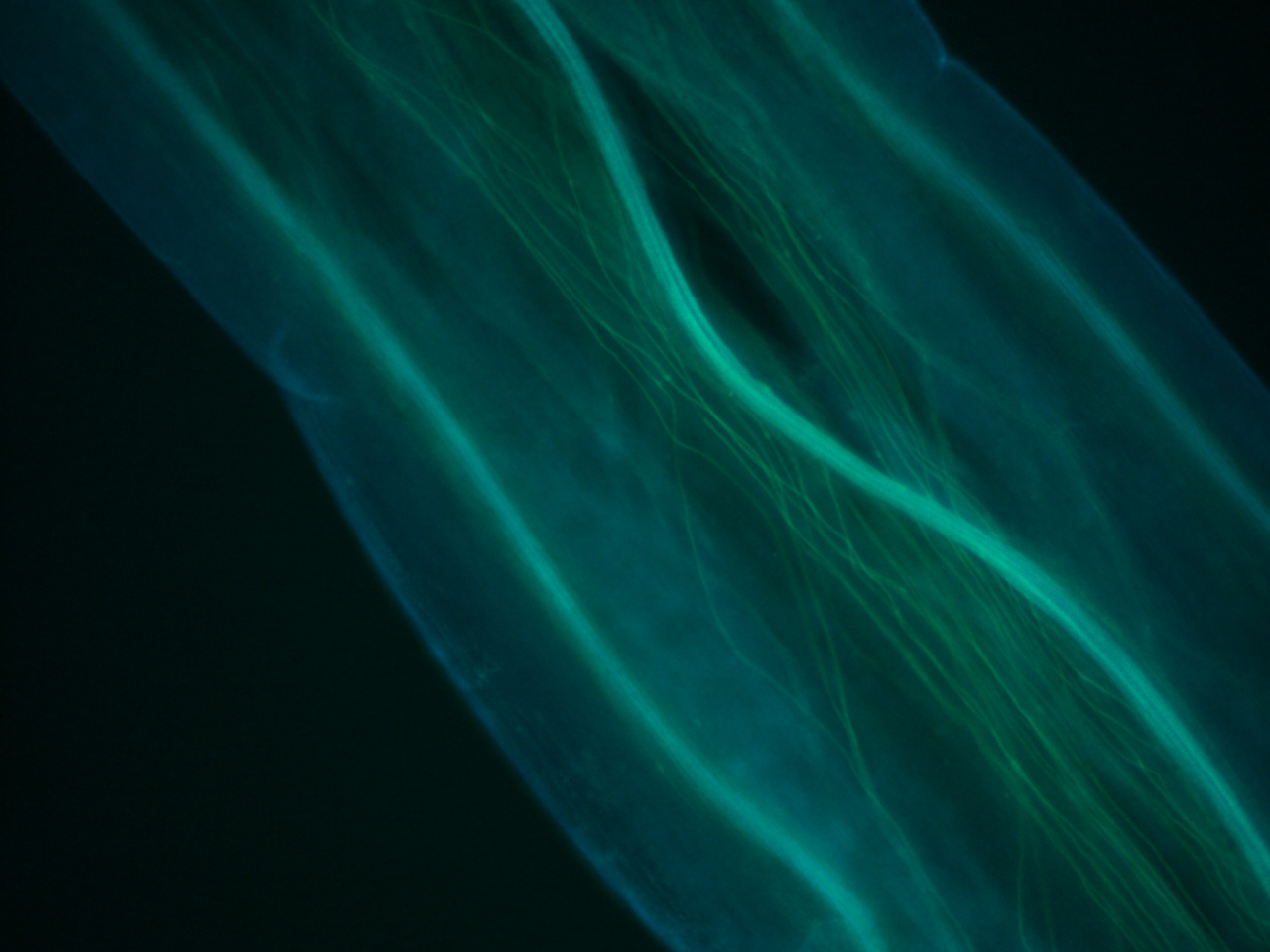
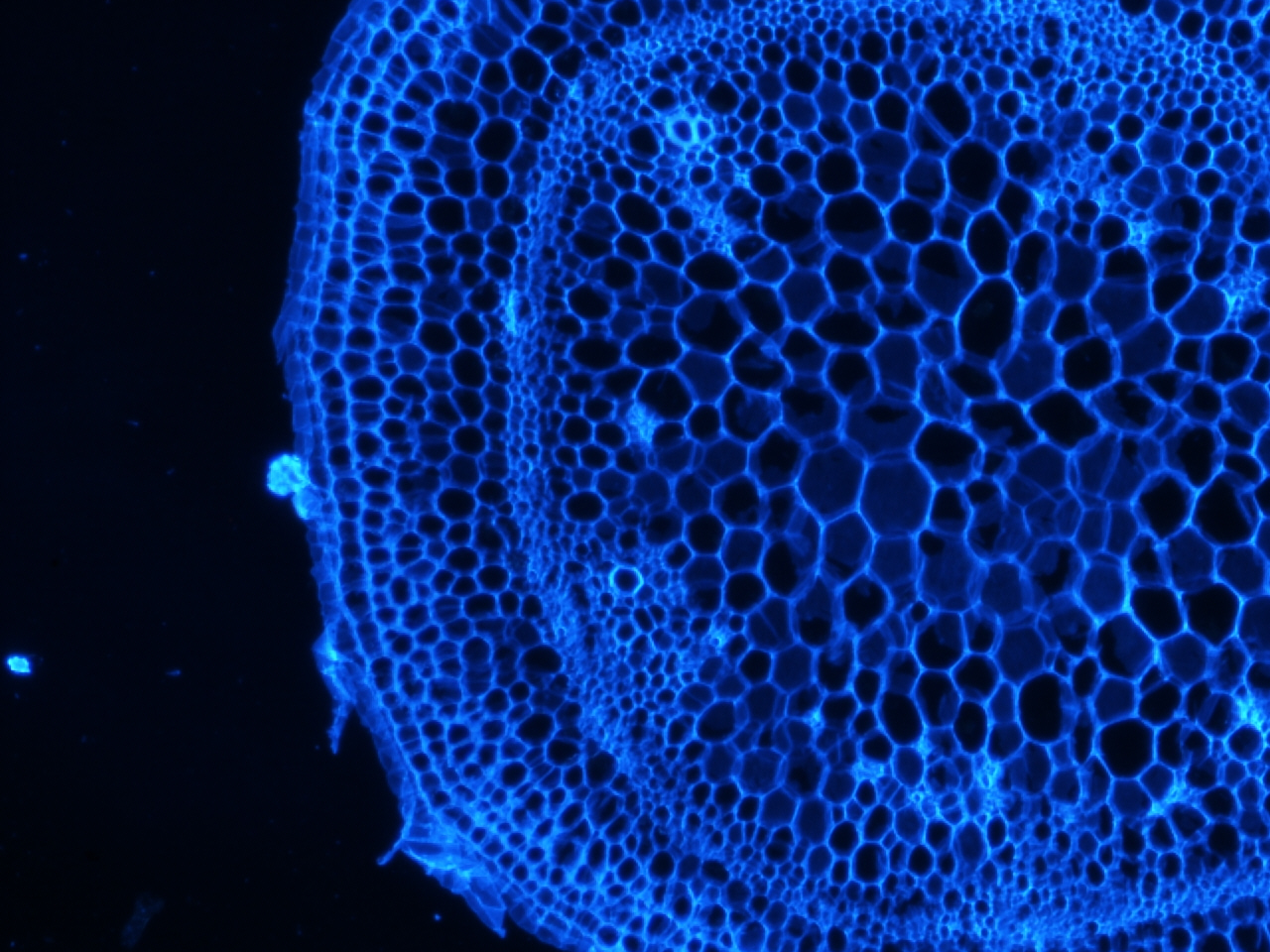
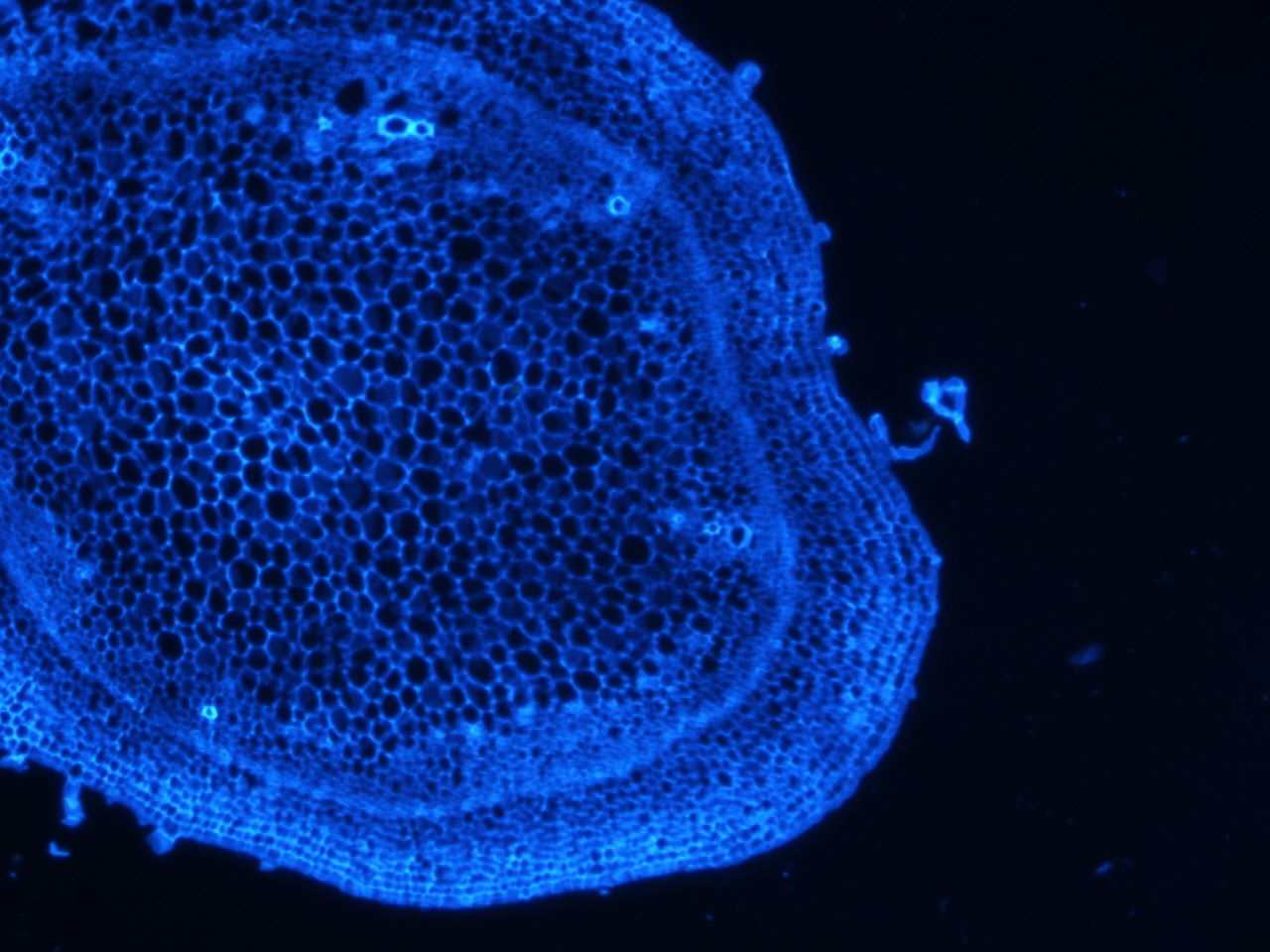
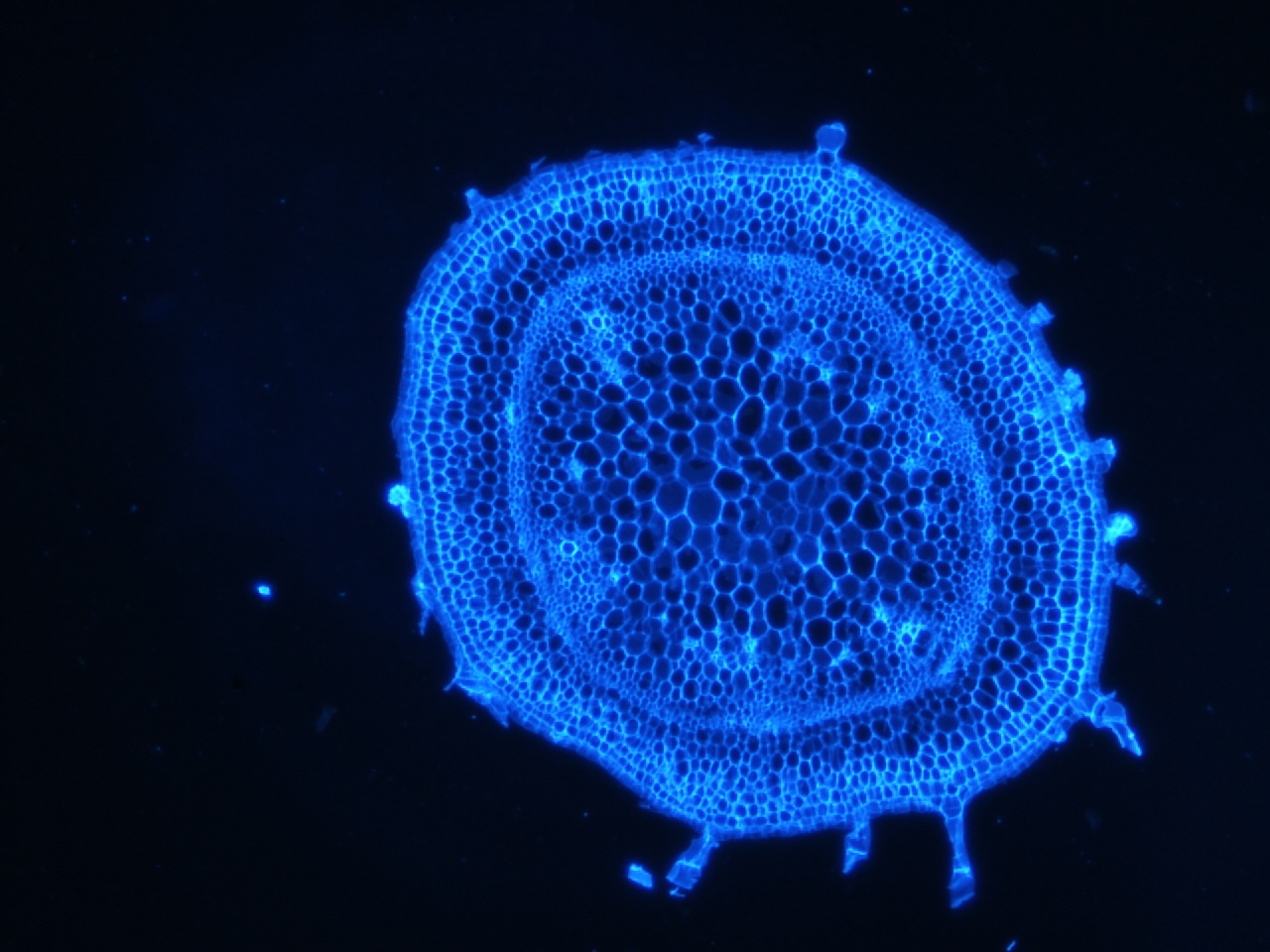
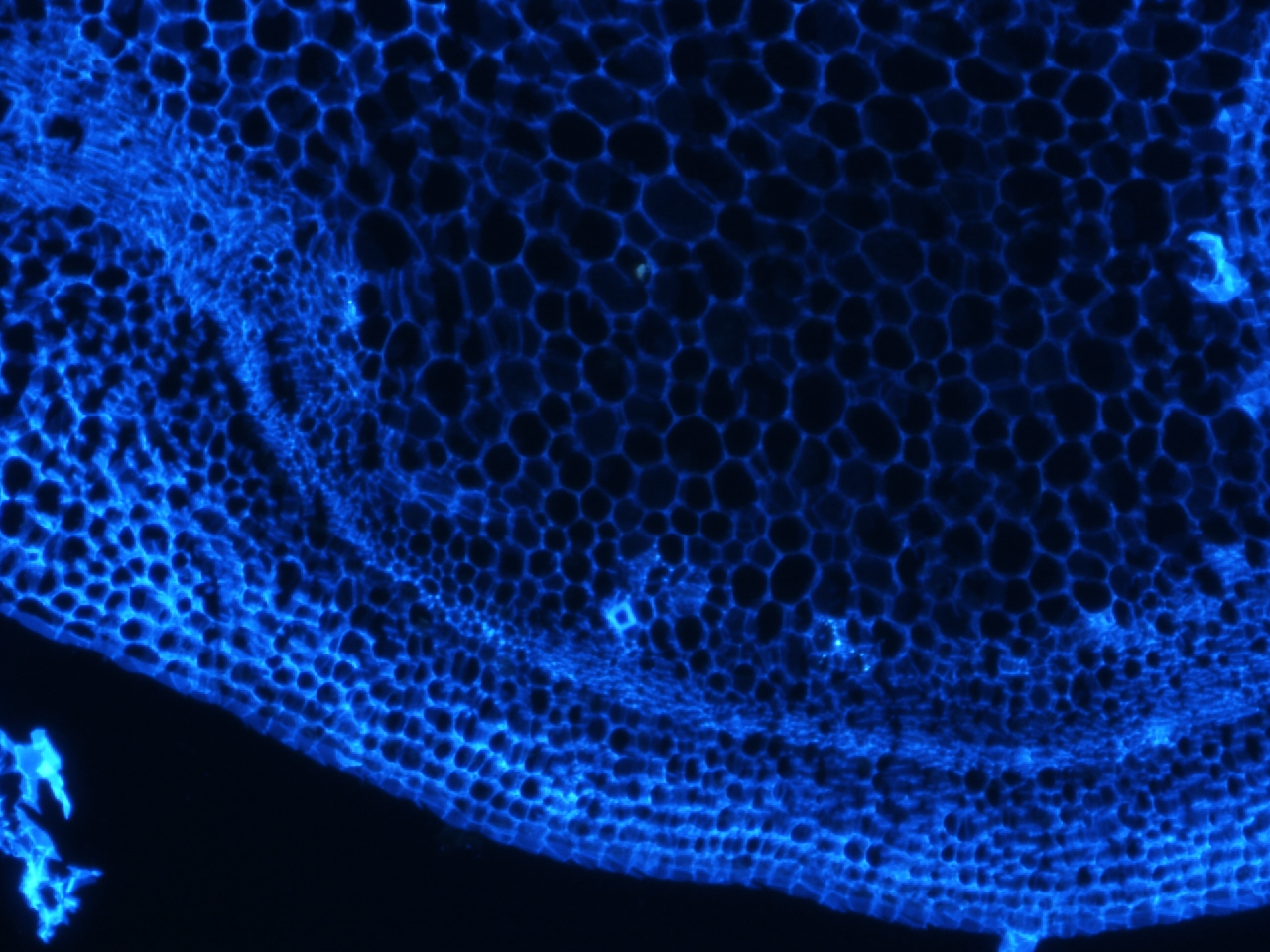
Stem stained with calcoflour
Calcoflour is used to visualize the cell walls of plants by staining a huge component of plant cell walls, cellulose.
I have been doing some histology of the stem and the shoot apical meristem in tomato, and recently could not resist staining a few of the slides with calcoflour since we just got the DAPI filter installed. I still have to get the stain timing and calcofluor dilution optimized, but if there is one thing I learned, it is that this stain gives an extremely strong signal using a very diluted amount.
Filter Set-up
Excitation Filter: 370/10 nm BrightLine® single-band bandpass filter
Dichroic Beamsplitter: 409 nm edge BrightLine® single-edge dichroic beamsplitter
Emission Filter: 409 nm blocking edge BrightLine® long-pass filter
Wavelength Display of all three filters
Thanks!
I am excited to analyze the results from all the samples that were collected this summer by my previous undergraduate intern Gillie Ish-Shalom and our lab technician Sharon Zimmerman. Also, special thanks to the Tomato Genetics Resource Center and Carlos de le Cruz from the Davis Microscope Facility for helping me set-up our microscope.
Resources
Bedinger Lab Tomato Pollen Staining Protocol free
This is the protocol I used. The only significant difference is I used plain ol’ Aniline Blue, not the Fluorochrome Aniline Blue.
Pollen and Stigma Structure and Function: The Role of Diversity in Pollination free
Control of pollen tube growth: role of ion gradients and fluxes free
comments powered by Disqus